Treat the Appendage, Improve Outcomes
Why Surgical Left Atrial Appendage Exclusion Matters in Atrial Fibrillation
The Clinical Problem: Afib-Related Stroke Remains a Major Cause of Morbidity
Atrial fibrillation (Afib) is a leading cause of ischemic stroke and is consistently associated with higher mortality, greater long-term disability, and increased healthcare utilization compared with non-Afib stroke. Contemporary epidemiologic reviews estimate that Afib accounts for approximately 20–30% of all ischemic strokes, with a disproportionate contribution to severe and fatal events.1,2
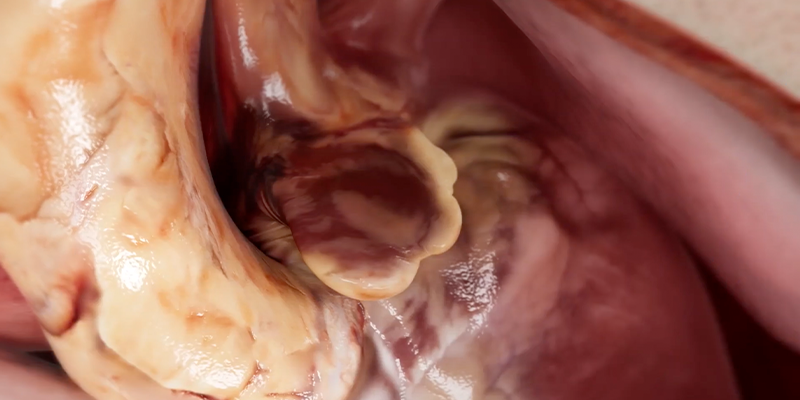
Mechanistic, autopsy, and multimodality imaging data demonstrate that in nonvalvular Afib, the vast majority of atrial thrombi, approximately 90%, originate in the left atrial appendage (LAA). This body of evidence establishes the LAA as the primary anatomic source of cardioembolism in this patient population.3,4
The Consequences of Leaving the Appendage Untreated
Oral anticoagulation (OAC) remains the cornerstone of stroke prevention in Afib. However, anticoagulation does not eliminate stroke risk and is frequently limited by bleeding complications, treatment discontinuation, and incomplete long-term adherence in real-world practice.5,6
From an economic perspective, Afib-associated stroke imposes a substantial and measurable burden on both payers and patients. Analyses of U.S. Medicare populations demonstrate that first-year healthcare costs following Afib-related ischemic stroke typically range from approximately $45,000 to $50,000 per beneficiary, with persistent cost elevation in subsequent years. In addition, device-detected or clinical Afib is associated with significantly higher annualized healthcare utilization and spending compared with patients without Afib.6,7
Evidence Supporting Surgical Left Atrial Appendage Exclusion
Randomized Evidence From Cardiac Surgery
The Left Atrial Appendage Occlusion Study III (LAAOS III) was a large, international randomized trial enrolling 4,811 patients with Afib undergoing cardiac surgery for other indications. Concomitant surgical LAA exclusion resulted in a 32% relative reduction in ischemic stroke or systemic embolism compared with no appendage treatment, on a background of guideline-directed anticoagulation. Importantly, this benefit was achieved without an increase in perioperative complications or mortality.8
Stroke Severity and Mechanism After LAA Exclusion
Beyond lowering the overall rate of stroke, LAA exclusion also changes the type and severity of strokes that occur when they do happen. In a prespecified post-hoc analysis of the LAAOS III trial, strokes among patients who underwent LAA exclusion were less likely to be cardioembolic, tended to be less severe, and were associated with lower 30-day mortality compared with strokes in patients whose appendage was not treated.9
These findings suggest that LAA exclusion not only reduces how often strokes occur, but also lessens their clinical impact when they do occur.
Surgical Technique and the Importance of Durable Closure
Long-term stroke protection depends on complete and durable LAA exclusion. In a large observational analysis published in The Journal of Thoracic and Cardiovascular Surgery, surgical LAA excision was associated with significantly fewer long-term ischemic strokes compared with intracardiac suture or extracardiac ligation techniques.10
Consistent findings have been reported in thoracoscopic series. Studies published in The Annals of Thoracic Surgery demonstrate that residual LAA stumps and incomplete closure are common mechanisms underlying residual thromboembolic risk. In these cohorts, thoracoscopic LAA clipping achieved higher rates of complete exclusion and shorter residual stump depth compared with stapled resection on follow-up imaging at approximately one year.11,12
The Electrical Role of the Left Atrial Appendage
In addition to its well-known role in stroke formation, the left atrial appendage has increasingly been recognized as an important source of abnormal electrical activity in atrial fibrillation, particularly in patients with persistent or more advanced disease. Electrophysiologic mapping studies have shown that the LAA can generate electrical triggers in a meaningful proportion of patients undergoing Afib ablation, and these triggers may help initiate or sustain Afib when pulmonary vein isolation alone is not sufficient.13
Mechanistic and computer-based modeling studies further support this concept, demonstrating that electrically isolating or surgically excluding the LAA in a way that disconnects its electrical activity can reduce abnormal reentry circuits and lower Afib recurrence in selected patients.14
Together, these findings suggest that the LAA is not simply a passive structure, but can actively contribute to atrial fibrillation. As a result, durable anatomic exclusion of the LAA may offer additional rhythm stabilization benefits when used as part of a comprehensive Afib treatment strategy in appropriately selected patients.
Conclusion: Comprehensive Afib Care Requires Excluding the Appendage
Across clinical trials and decades of research, evidence consistently shows that most strokes related to non‑valvular atrial fibrillation originate in the LAA.1,2,3,8,9 Surgically excluding the LAA has been shown to provide lasting stroke protection, reduce the severity of strokes when they do occur, and, based on growing mechanistic and electrophysiologic evidence, may also help stabilize heart rhythm in selected patients as part of comprehensive atrial fibrillation care.8,9,13,14
Emerging data suggest that prophylactic LAA exclusion during cardiac surgery may reduce stroke risk in patients at high risk of developing atrial fibrillation, sometimes referred to as ‘pre-Afib.’ This concept, highlighted by Dr. McCarthy and Ferguson in their 2025 Mini LeAAPS paper, is promising but requires further evidence before it can be confirmed.
Taken together, these findings support routine, complete, and confirmed LAA exclusion as a key component of atrial fibrillation management in patients undergoing cardiac surgery.